Determination
of Absolute Zero: Charles's Law
Objective: Determine absolute zero from extrapolation of
volume and temperature data using Charles's Law
Materials and Procedures: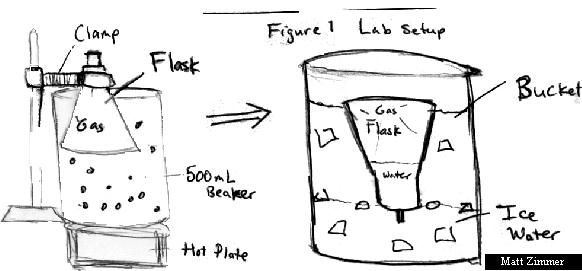
1. Prepare a cold water bath using a large
bucket approximately 3/4 full.
2. Fit the dry 125 mL Erlenmeyer flask with a one-hole stopper.
3. Prepare a hot water bath on
the hot plate in a 500 mL beaker.
4. Clamp the Erlenmeyer flask in
the hot water bath such that most of it is under water.
5. Allow the water to boil 5 min
to allow the zeroth law of thermodynamics to
take effect.
6. Record the temperature of the
boiling water to 0.5o C.
7. Place the solid glass rod into
the one hole stopper, then remove the
Erlenmeyer flask from the beaker.
Allow it to cool for 2-3 minutes until cool to the touch
8. Invert the Erlenmeyer flask
and place it in the cold water bath.
Remove the glass rod while under water and still inverted, then totally
submerge the inverted flask for 5 min.
9. Stir occasionally. Record the temperature of the ice bath to 0.5o
C just prior to removing the Erlenmeyer flask.
The
next step should be done as quickly as possible to avoid warming of the flask.
10. Using your finger tips, raise
the inverted flask until the water level inside and outside the flask are
equal. Now seal the inverted flask with
the glass rod.
11. Determine the volume of water
collected in the inverted flask, then the volume of the entire flask. This will allow you to calculate the volume
of the air at both temperatures.
Analysis
1. Plot the volume (y-axis) of
the air in the flask vs. oC
temperature. Allow room on your graph to
extrapolate back to zero volume on the y-axis such that the absolute
zero temperature can be determined.
2. In your analysis, respond to
the following questions:
a) Why did the water flow into the flask when inverted in the cold water
bath? Explain in terms of kinetic theory.
b) Why was it necessary to allow the two water levels to become equal in
Step 10?
c) From your graph, what is the predicted temperature for zero volume of
a gas. Calculate a % error.
d) Explain why the temperature cannot drop below absolute zero.
e) What happens to air before it gets close to absolute zero that would
prevent an accurate determination of absolute zero?
f) Theoretically, what happens to molecules at absolute zero? How could this be performed for better
results?
g) Explain Charles’s Law
h) How could this lab be designed to be more accurate?
Back
to the Brockport High School Science Department
