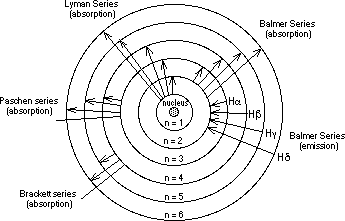
Obj: Determine photon energy and quantum number of electrons returning to n = 2 in the hydrogen atom.
Energy levels of Hydrogen Atom:
The Balmer Series
Obj: Determine photon energy and quantum number of
electrons returning to n = 2 in the hydrogen atom.
Materials: H2 gas tube, spectrometer
Methods & Analysis
(Note: run the spectrum tube for no more than 30 seconds with
a 30 seconds cooling period.)
1. Looking through the diffraction grating at the
ionized hydrogen gas, estimate the λ of the red, blue, and
violet bright lines to the nearest 10 nm using the spectrum
chart. [1 nm = 1 x 10-9 m] There should be a minimum
of three lines visible.
2. Calculate for each color the energy E (hc/λ)
of that photon in Joules and eV.
3. Compare your energy calculations with the
energy differences shown on your reference table.
Account for any discrepancies.
What do these energy differences indicate?
4. For each visible photon, determine the quantum
number n from which it returned to n = 2 of hydrogen.
Use your reference table for assistance.
5. Which color has the highest frequency?;
energy?; the
shortest λ ?; most and least momentum?
6. Repeat steps 1-5 for mercury gas, choosing two
colors.
Back to the Brockport High School Science Department